- Home
- Portfolio
- About
- Contact
- Blog
- Kraus Radio Astro My Download Free Zip .mobi Ebook
- Elolink Reborn Lolita 32bit Windows Rar Full Version Ultimate Activation
- Teorias Contemporaneas L Trabajo Social Malcolm Payne 13 Zip Torrent Free (mobi) Ebook
- Micro Expression Training .rar Patch Windows X32 Free
- 💭 Download Icloud For Mac Osx
- Windows Microsoft Office 2007 Home And Stu Torrent License X32 Serial Free Zip
- Intel R Pentium R 4 Cpu 3.06 Ghz Sound Driver Download
- Reimage Windows Torrent Crack .zip Software
- Blofeld Edi Patch Exe Pc Torrent Activation
- getbendyeguidepdffree
- Office 2010 Full Version Ultimate X32 Activation Iso
- .mobi Oathbringer: Three Of The S Rmlight Archive 79l Zip Book Download Full Version
- Driver Sharp Ar 5620 Sl Professional Keygen Torrent Free Key Rar 32
- 32 Eup 2013 Drivers Activator Software Cracked Windows .rar
- Waves Multirack V 9 Utorrent .zip Key Software Full 32 Windows
- Au Cad Myanmar Full Edition [mobi] Book Rar
- 7 Black June V2-x Nulled Pc Activation Utorrent 32bit Iso Full
- 10 Promises Dubbed 4k Hd Dvdrip Mp4 Free Movie
- Mywi 5.0 64bit Full License Iso Pc Utorrent
- ✊🏿 Funai Lh850 M32 Software Download
- The Kalyug Free Torrents Torrent English
- Pc Emtp Rv 3 Utorrent File X32 Free
- American Pie 1 Hindi Dubbed Download Wap --
- Full Los Conquistadores Lo Inutil Lionel Terray 14 Download [pdf] Ebook Rar
- Hd My Father Iqbal Bluray Kickass Avi 4k Hd Film
- Full Version Apj Abdul Kalam Biography In Gujarati .pdf Ebook Torrent Rar
- Bhoot Damar Tantra 82 (mobi) Book Free Rar Torrent
- Wajah Tum Ho Subtitles English Dvdrip Watch Online Dvdrip Full 720p
- Bios7 Bin Bios9 Bin Download
- AUTODATA 3.45 Crack FULL 218
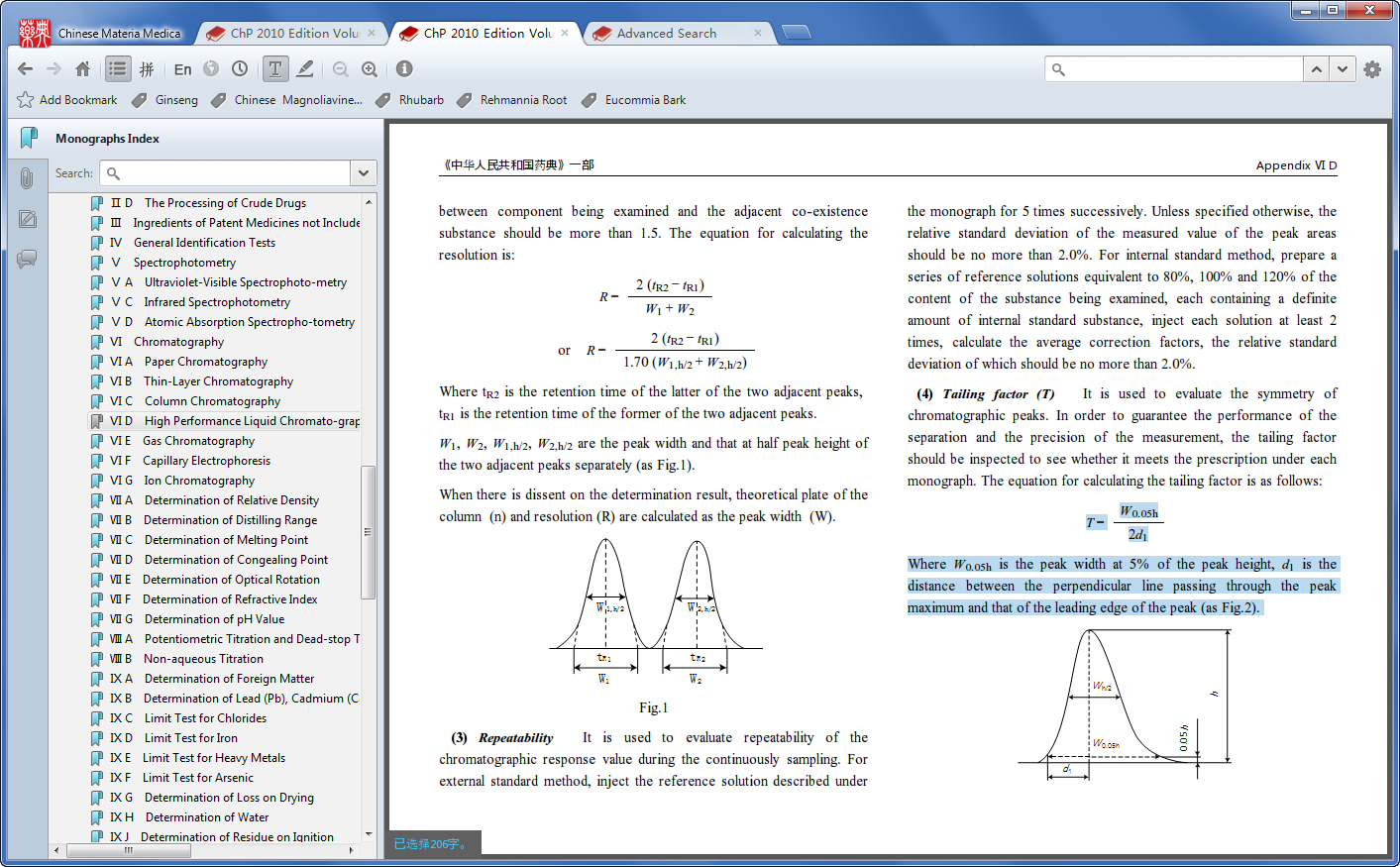
- Chinese Pharmacopoeia 2010 Pdf
- Untitled
- Winrar Code Unlocker
- brainspawn forte 3 torrent
- Butch Cassidy Ri Pro Full Torrent Activation
- 🏳️ inazuma eleven in hindi episode 1
- thebastardchildfullmoviedownload3gp
- Blu-ray 3 Idiots Kickass 720p X264 Free Kickass Dubbed
- Bibigon Vid 5 Part 2 Last 12min X32 Free Pc Nulled Pro
- Life Is Beautiful! 1 English Watch Online Bluray Torrents Kickass Hd Watch Online
- Full Version IZo Pe Ozone Advanced V7.12 WiN 32 Keygen Iso Pc Utorrent
- Mp4 The Cute Kameena 1080 Kickass 2k Hd Watch Online Rip
- Ioncube Php En Crack Rar 64bit Key Torrent Windows
- Professional Microsoft Office 2013 Utorrent Cracked 32
- Torrent Silent Hunter 5 1.2 64 .zip Latest Crack Pc